In IVF ICSI, What could cause embryos to stop developing
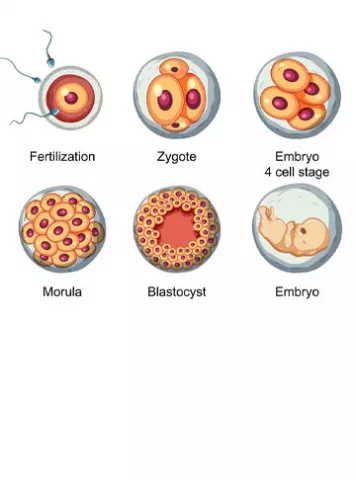
In IVF /ICSI it is common for the embryos to stop developing in the lab. It is called embryonic arrest and it can happen at any stage: some embryos do not reach past single cell/ zygote stage and few undergo arrest during the cell division process or during cleavage stage or any time before they reach the stage of blastocyst.
Embryonic arrest can be of natural causes or may be due to human errors or due to suboptimal lab conditions.
The genetic material from the mother and father is carried by an embryo and this genetic information is needed for the single celled embryo to grow into a baby.
Oocyte factors
In the beginning stages up to 4-8 celled stage, it is only the genetic information of the oocyte which is responsible for cell division. It is only after this stage, the embryonic genome (the first cell of the human life) gets activated and further development takes place. When this genome activation fails, there occurs arrest of embryo.
Studies have shown that embryonic arrest may be a consequence of the failure to activate important genes needed for development and/or also due to stress caused by the environment/ culture conditions (temperature, humidity, pH, gases, etc.), the oxidative stress that the embryos are exposed to and the quantity and distribution of their cellular components.
It can be said that embryo arrest in-vitro is a useful biological mechanism to rule out the embryos that do not achieve certain quality criteria.
Around 50-70% of embryos do not grow to the stage of blastocyst (Wong et al. 2010) and nearly 40% of all the patients, will have one arrested embryo per cycle (Betts et al. 2008).
After sperm fertilizes the egg and forms the embryo, it forms the genome of the embryo. This will have embryo’s complete set of DNA and with all the instructions for its life.
The embryo’s genome is not activated until about Day 3, when the embryo has about 8 cells (Hanna et al. 2018). Till then, the embryo depends on stored maternal factors in the oocyte. Since the embryo’s DNA is not active yet, it cannot make its own factors and embryo depends on stored maternal factors for development which are present in the oocyte.
These factors play important roles in:
- Unpacking the DNA of sperm.
- Combining the DNA of the oocyte and the sperm to form the embryo’s genome
- Turning off the oocyte or sperm genetic programs and switching on the embryonic program
- Copying of DNA and undergoing 3 rounds of cell division (to reach the 8 celled stage)
The oocyte has stored RNAs that are translated into proteins.
Some of the maternal RNAs (and the corresponding gene/protein) which can be found in the early embryo are (Wong et al. 2010) those involved in cell division (ANLN, AURKA, CFL1, ECT2, and FGFR1), Energy production (ATP2C1), Transcription of DNA to mRNA (TAF4, TBP), Processing of RNA (CPEB1, PARN, SYMPK and DICER1) and DNA reprogramming (EHMT2, DNMT3B).
Wong et al. (2010) studied these maternal RNAs and they found that transcripts involved in cell division and processing of RNA were all reduced in arrested embryos as compared to normal embryos.
The subcortical maternal complex (SCMC): It’s a multi-protein complex –with protein molecules like KHDC3L, NLRP2/5/7, OOEP, PADI6, and TLE6- which plays important role in
- Positioning the spindle (involved in separation of chromosomes during cell division)
- Controlling which maternal RNAs are used by the oocyte
- Localizing mitochondria to important areas of the cell where energy is needed
- Reprogramming the sperm/egg DNA to the embryo DNA program
Few studies on these protein molecules have shown that mutations in these, can lead to embryonic arrest. (Bebbere et al. 2016).
Xu et al. (2016) did their study regarding PADI6 molecule and found that -One patient with mutation of PAD16, had 6 cycles resulting in 35 oocytes, out of which 22 fertilized and all arrested on day 3.
Alazami et al. (2015), in their study on patients with mutation TLE6, found that -2 patients had a total of 5 cycles leading to 58 oocytes, among them 3 fertilized and got arrested at the 1-4 cell stage.
Wang et al. (2018) in their study on patients with mutation of KHDC3L, found that one patient had two cycles resulting in 19 oocytes, out of which 15 fertilized but all arrested on day 5/6, at the morula stage.
Though these mutations are rare and may not affect many women, it’s evident that any disruption in SCMC protein complex can lead to embryo arrest.
Aneuploidy:
Arrested embryos have a tendency to be aneuploid. Qi et al. (2014) in their study on arrested cleavage-stage embryos, found that almost 98% of them were having aneuploidy. In the study it was also found that arrested embryos had more “complex” abnormalities where multiple chromosomes were affected.
In another study, it was found that around 70% of arrested embryos were having aneuploidy. But they had looked at only 5 chromosomes out of the 23 chromosomes, during the PGS study. It may seem that the numbers might have been higher if all the chromosomes were studied (with newer technology). (Maurer et al. 2014)
Maternal Age:
Oocytes of advanced aged patients have higher risk of chromosomal defects, in such cases there occurs growth arrest as a natural defense mechanism to prevent the birth of a baby with genetic defect.
Mitochondria:
Mitochondria are the powerhouse of the cell, as they generate most of the cells-supply of ATP, the energy molecule. As women age advances, mitochondria start to deteriorate and this causes errors in chromosome separation or result in aneuploidy.
In a study on pigs, researchers found that poor quality oocytes, have low levels of mitochondria. Embryos generated from these oocytes were prone to embryo arrest (Cagnone et al. 2016). In the study, mitochondria were injected into these oocytes to increase to normal levels. Lesser numbers of embryos arrested and more reached blastocyst stage.
Reactive oxygen species (ROS):
ROS can result in DNA damage and this can cause embryo arrest in different ways.
Telomeres are protective repetitive DNA sequences at the end of every chromosome.
ROS cause the telomere DNA to shorten, which can lead to cellular senescence.
Studies have shown that shortened or damaged telomeres can cause embryo arrest (Betts et al. 2008).
Sperm factors:
Embryos inherit the centrosome from the sperm. Centrosomes have a role in separating chromosomes during cell division. The centrosome from damaged sperm may get faulty in separating chromosomes and this could result in embryo arrest.
DNA fragmentation
Studies have shown that sperm DNA fragmentation (SDF) has been associated with decreased chances of blastocyst formation and significant damage could cause the embryo to arrest. (Sedo et al. 2017).
Laboratory factors:
Other major reason for embryonic arrest is absence of optimal culture conditions, infected culture medium or malfunctioning incubator. There is hindrance of cell division and cells stop growing when there is suboptimal conditions in the lab.
Environment and air of IVF labs need to be clean and free from any pollutants.
Volatile organic compounds (VOCs) like Ethanol, isopropyl alcohol (for decontaminating surfaces), Propene, acetonitrile, styrene (from plastics), Formaldehyde, acetaldehyde (found in paints, gasoline, smog) are molecules in the air that can be harmful to embryos. Some VOCs are known carcinogens and can result in DNA damage. VOCs have been linked to embryo arrest or failure to develop blastocysts as well as reduced overall success (Mortimer et al. 2018).
It’s essential to have a clean lab and clean air to protect the embryo health.
Articles
2022


Guide to infertility treatments Blastocyst Culture And Transfer
What Is The Best Advice After Embryo Transfer
In Vitro Fertilisation, popularly referred as IVF has captured attention since...
2022


Guide to infertility treatments Blastocyst Culture And Transfer
Elective single embryo transfer (eSET): A safe option in IVF
What is Single Embryo transfer or SET? Single embryo transfer (SET)is an el...
2022


Guide to infertility treatments Blastocyst Culture And Transfer
Do’s and Dont’s Post Embryo Transfer
When a couple undergoes IVF or test tube baby treatment, they are in a turmoil...
2022


Guide to infertility treatments Blastocyst Culture And Transfer
Can Single Embryo Transfer Result in Twins?
As the quote by Josh billing “there are two things in life for which we are ...
2022


Guide to infertility treatments Blastocyst Culture And Transfer
Single or Multiple Embryo Transfer – Necessary Points to Know
Embryo Transfer Even though science can give a lot of options, it is not ne...
2020


Guide to infertility treatments Blastocyst Culture And Transfer
What To Do After Embryo Transfer To Increase Success?
For many couples, the path to parenthood is not always how they may have envis...
2020


Guide to infertility treatments Blastocyst Culture And Transfer
What is the diet i require to take after the embryo transfer?
In vitro fertilization is one of the most advanced techniques in the field of ...
2020


Guide to infertility treatments Blastocyst Culture And Transfer
Spotting After Embryo Transfer – Am I going to Miscarry?
Infertility is a condition that affects one in ten couples who are trying to c...

Blastocyst Culture And Transfer
फ्रोजन भ्रूण प्रत्यारोपण (ब्लास्टोसिस्ट) प्रक्रिया
किसी ने ठीक ही कहा है जब भी मैं ...
Pregnancy Calculator Tools for Confident and Stress-Free Pregnancy Planning
Get quick understanding of your fertility cycle and accordingly make a schedule to track it